Glowing Recommendations
New resource helps researchers select fluorescent proteins for experiments
- 5 minute read
- Feature
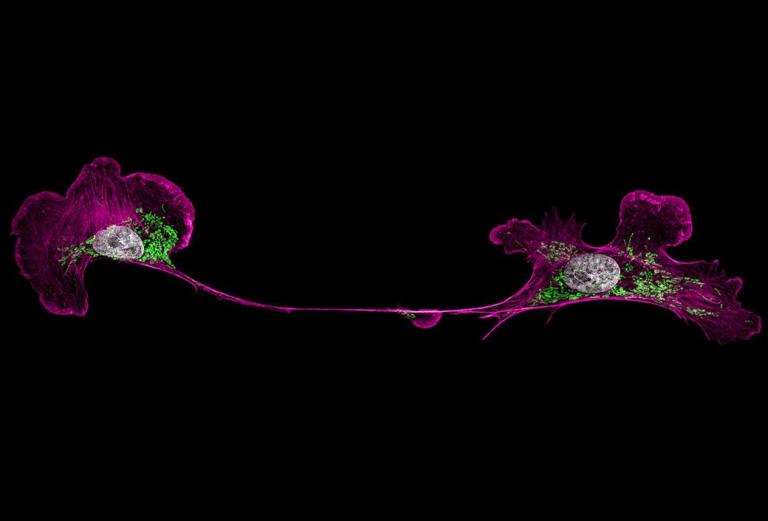
Since green fluorescent protein, originally discovered in jellyfish, was adapted for scientific research in the 1990s, dozens upon dozens of fluorescent proteins—the tiny, glowing workhorses of biomedical imaging that allow researchers to observe gene activity in a cell, organ or whole organism—have flooded the market, in a rainbow of colors and with a range of additional essential properties.
Researchers designing fluorescence microscopy experiments across the HMS community enjoy a buffet of options and the breadth of choices creates a conundrum: Which proteins are best suited to their projects?
“It’s an extremely common question,” says Talley Lambert, an HMS research associate in cell biology and manager of the Cell Biology Microscopy Facility at the School. “You want to know which is right before you spend three weeks fusing a protein to your gene of interest.”
Because not everyone has the training to understand all the factors that distinguish one fluorescent protein from another, it can be difficult to choose the correct fluorophore, one that will not hamper experiments or confound results. Compounding the problem, there has not been a central repository for those who wanted to look up such details.
Now there is. Lambert has built a tool to help researchers compare and select the best fluorescent proteins for individual experiments: the Fluorescent Protein Database, or FPbase.
The searchable web-based application aggregates information about fluorescent proteins from multiple sources and delivers them to one place in user-friendly formats.
Although FPbase doesn’t replace the value of Quad researchers’ consulting with Lambert, facility director Jennifer Waters, or the facility’s team, its centralization and visual nature can aid those discussions.
“When I get questions, I can’t just say ‘go to the site,’” says Lambert, “but it helps me demonstrate to people what’s best.”
Even those who’ve done their homework are likely to have further questions, he adds. Then there are “all these caveats and potential misinterpretations” that can arise when users make choices based on a limited understanding of fluorescent-protein characteristics.
“My fear is—because these proteins are so complicated, and some properties are easier to measure than others—that users will draw premature conclusions from a quick glance at one property or their experiment won’t work for some other reason,” says Lambert.
Extra credit
Lambert began playing with the FPbase concept as a sort of extracurricular activity.
“It wasn’t specifically required of me,” he says, “but how to select a fluorescent protein is such a common question that I wanted to see if I could build something that would save people time and maybe contribute to reproducibility.”
On a personal level, Lambert enjoyed learning new programming and layout skills, including a crash course in Django, a Python-based web design tool.
“It was a fun and gentle introduction to data science,” he says.
Lambert wrote scripts to aggregate smaller existing lists of fluorescent proteins; created a schema to define the properties of each protein, validate them, and put them into a consistent format; and made sure the results were “easily consumable.”
FPbase went live this spring.
“I was scared to release it,” Lambert confesses, “but the response so far has been positive. We’ve received emails with good suggestions for additions.”
The database receives around 900 hits per day. So far, most users have come from outside HMS.
Lambert and Waters think the interest beyond HMS is the result of their having talked about FPbase on Twitter. They want to make sure the Quad community also knows the resource is available.
One-stop shopping
Using FPbase, researchers can explore—and set constraints for—the spectra that fluorescent proteins absorb and emit and other characteristics such as brightness, acid sensitivity, photostability (how long a protein can withstand light exposure), quantum yield (how efficiently it converts absorbed light into emitted light), and lifetime in nanoseconds. Each data set provides references to original publications about the proteins.
FPbase also includes a feature that allows researchers to enter key attributes of the microscopes in their labs, such as lasers, filter sets, and cameras, to help scientists determine how well different fluorescent proteins will work with particular instruments, says Waters, who is also an HMS lecturer on cell biology.
In addition to its comprehensiveness and usability, FPbase is the only fluorescent protein database designed to be editable by community members.
“Users can upload information into the database, allowing it to grow and stay current without any one person or group needing to maintain it,” Waters explains.
“If something isn’t satisfying, you can make that part of the website better for yourself and others,” adds Lambert. “You can update and edit if you find mistakes.”
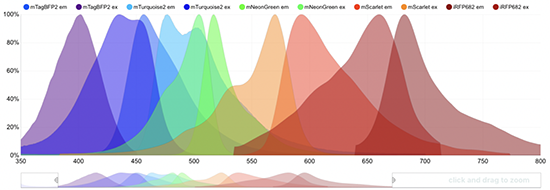
Lambert moderates user-contributed content to ensure that people add to FPbase without “messing it up,” he says. Users must provide citations, and he can make corrections or fill in missing information.
“I hope people use FPbase not just to consume information but to contribute,” he says. “I want it to take on a life of its own.”
Stephanie Dutchen is a science writer in the HMS Office of Communications and External Relations.
Images: Cell Biology Microscopy Facility (top and middle); Talley Lambe